Presenilin 2 loop region, Rabbit Polyclonal Antibody
As low as
US$327.00
Only %1 left
Catalog Number
R-1681
- Product Name Presenilin 2 loop region, Rabbit Polyclonal Antibody
- Product Description Rabbit anti-Presenilin 2 loop region Polyclonal Antibody (Unconjugated), suitable for WB, ICC.
- Alternative Names AD3LP, AD5, E5-1, STM-2
- Application(s) ICC, WB
- Antibody Host Rabbit
- Antibody Type Polyclonal
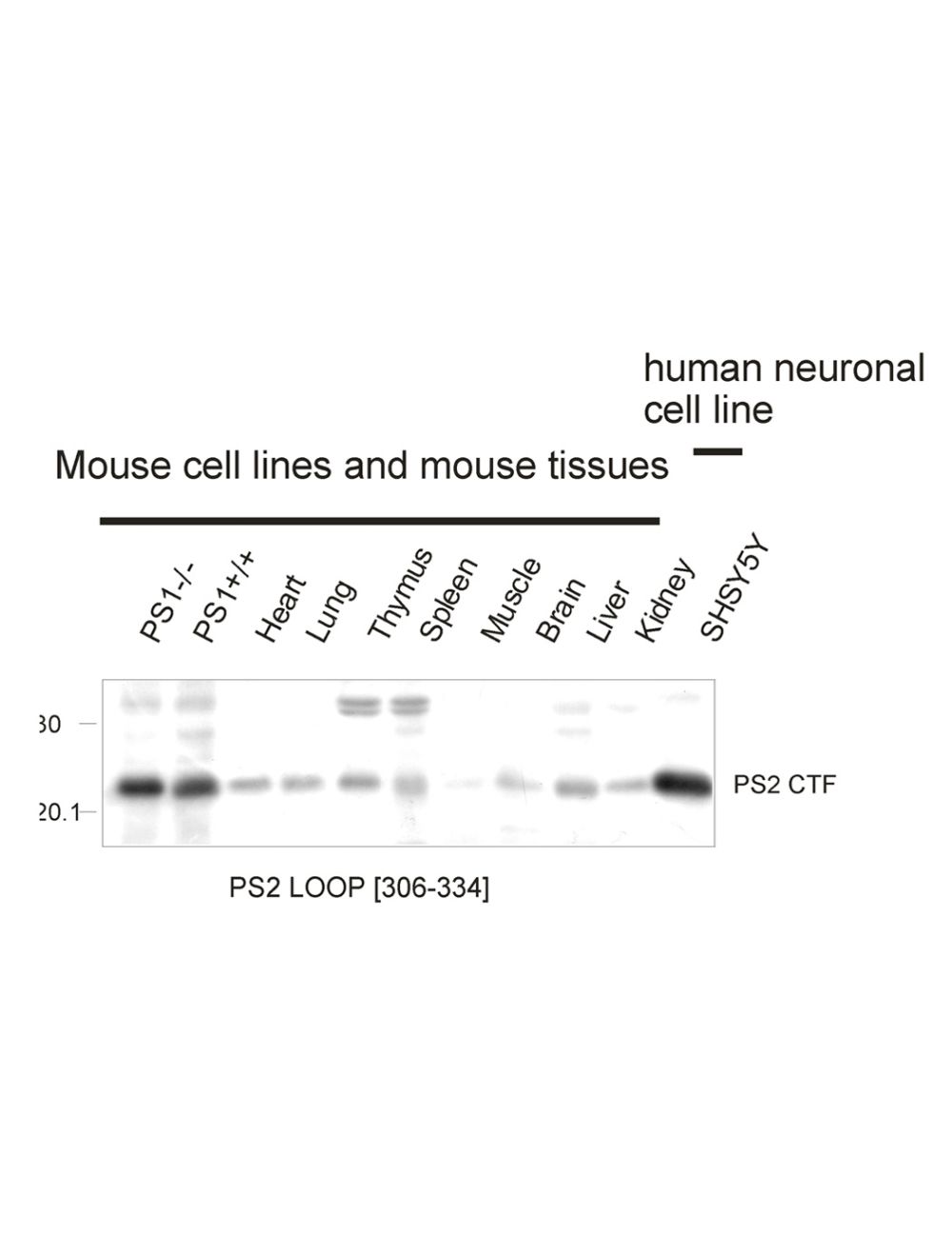
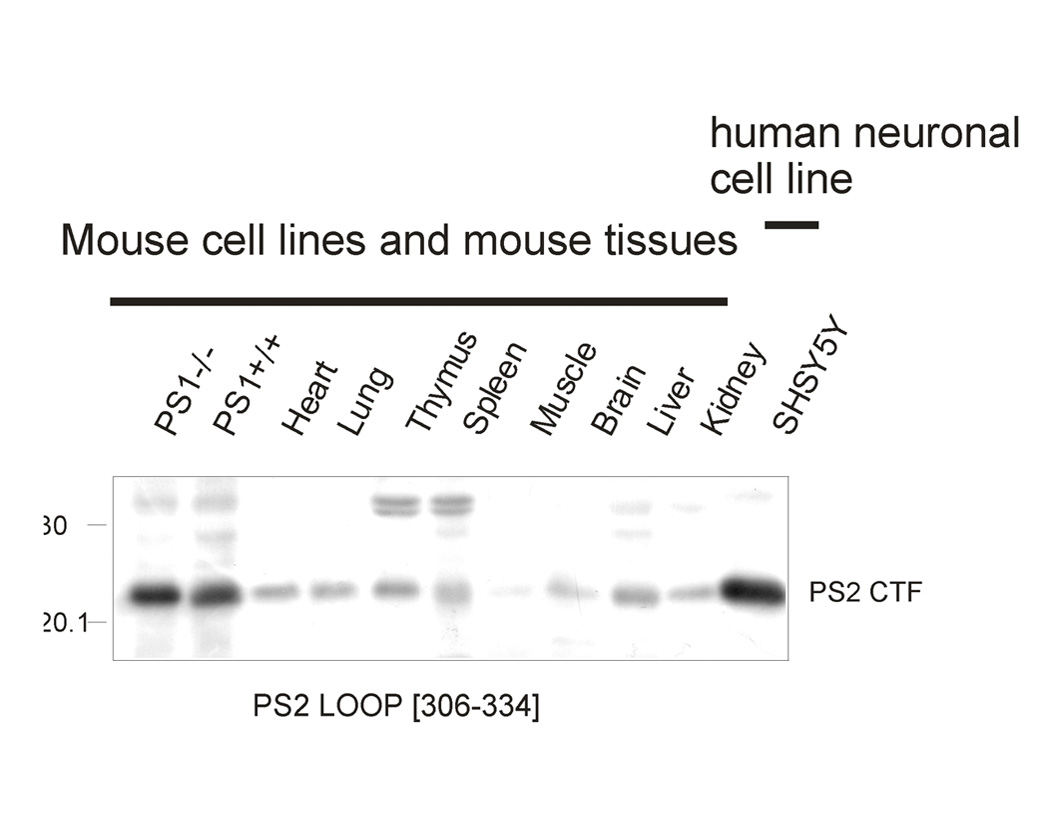
- Specificity Confirmed by Western blot using mouse and human brain and knock down of presenilin 2 in vitro using siRNA see ref 6 below. Not reactive with presenilin 1.
- Species Reactivity Human, Mouse
- Immunogen Description A synthetic peptide (KLDPSSQGALQLPYDPEMEEDSYDSFGEP-C) corresponding to human PS1 [306-334] in the loop region conjugated via additional C-terminal Cys to Diphtheria toxoid.
- Conjugate Unconjugated
- Purity Description Protein G purified IgG
- Regulatory Status For research use only.
Product Info
- Product Description Rabbit anti-Presenilin 2 loop region Polyclonal Antibody (Unconjugated), suitable for WB, ICC.
- Application(s) ICC, WB
- Application Details WB and IP. Suggested dilution of 1:1,000 is recommended for WB. Full length presenilin 2 (448 aa) has relative MW of about 45 kDa, with this antibody most commonly detected as cleaved CTF of 22 kDa with this antibody. Human or mouse brain samples commonly prepared with reducing agent (50mM DTT), urea (2.3 M), SDS (1.5%) in 62.5 mM Tris-HCL pH 6.8 sample buffer (without boiling) heating to 50 C for 15 min. The suggested dilution for IP is 1:100 . Biosensis recommends that the optimal working dilution should be determined by the end user.
- Target Presenilin 2 loop region
- Specificity Confirmed by Western blot using mouse and human brain and knock down of presenilin 2 in vitro using siRNA see ref 6 below. Not reactive with presenilin 1.
- Target Host Species Human
- Species Reactivity Human, Mouse
- Antibody Host Rabbit
- Antibody Type Polyclonal
- Antibody Isotype IgG
- Conjugate Unconjugated
- Immunogen Description A synthetic peptide (KLDPSSQGALQLPYDPEMEEDSYDSFGEP-C) corresponding to human PS1 [306-334] in the loop region conjugated via additional C-terminal Cys to Diphtheria toxoid.
- Purity Description Protein G purified IgG
- Format Lyophilized from PBS, pH 7.4. Contains no preservative.
- Reconstitution Instructions Spin vial briefly before opening. Reconstitute in 500 µL sterile-filtered 1X PBS, pH 7.2-7.6. Centrifuge to remove any insoluble material.
- Storage Instructions Short term storage at 2-8°C for one week. At -20°C as an undiluted liquid for up to 12 months.
- Batch Number Please see item label.
- Expiration Date 12 months after date of receipt (unopened vial).
- Alternative Names AD3LP, AD5, E5-1, STM-2
- Uniprot Number P49810
- Uniprot Number/Name P49810 (PSN2_HUMAN)
- Scientific Background Autosomal dominant mutations in presenilin 2 are the second major cause of early-onset familial Alzheimer's disease. Presenilin 2 is a multi-transmembrane protein which undergoes endoprotelysis to form an N-terminal fragment of about 29 kDa and C-terminal fragment of about 22 kDa. Presenilin 2 forms the catalytic core of the gamma-secretase complex which cleaves type 1 transmembrane proteins including the amyloid precursor protein to generate the C-terminus of the amyloid beta peptide.
- Shipping Temperature 25°C (ambient)
- UNSPSC CODE 41116161
- Regulatory Status For research use only.
Specifications
-
General References
Culvenor, J.G., Ilaya, N.T., Ryan, M.T., Canterford, L., Hoke, D., Williamson, N.A., McLean, C.A., Masters, C.L., and Evin, G. (2004) Characterization of Presenilin complex from mouse and human brain using Blue Native gel electrophoresis reveals high expression in embryonic brain and minimal change in complex mobility with Presenilin mutations. Eur. J. Biochem. 271, 375-385.
Ilaya, N.T., Evin, G., Masters, C.L., and Culvenor, J.G. (2004) Nicastrin expression in mouse peripheral tissues is not co-ordinated with Presenilin and is high in muscle. J. Neurochem. 91, 230-237.
Beher, D., Fricker, M., Nadin, A., Clarke, E.E., Wrigley, J.D.J., Li, Y.-M., CULVENOR, J.G., Masters, C.L., Harrison, T., and Shearman, M.S. (2003) In vitro Characterization of the Presenilin-dependent _-secretase complex using a novel affinity ligand. Biochem. 42, 8133-8142.
Evin, G., Smith, M.J., Tziotis, A., McLean, C., Canterford, L., Sharples, R.A., Cappai, R., Weidemann, A., Beyreuther, K., Cotton, R.G.H., Masters, C.L., and Culvenor, J.G. (2002) Alternative transcripts of Presenilin-1 associated with Frontotemporal Dementia. NeuroReport 13, 917-921.
Evin, G., Sharples, R.A., Weidemann, A., Reinhard, F.B.M., Carbone, V., Culvenor, J.G., Holsinger, R.M.D., Sernee, M.F., Beyreuther, K., and Masters, C.L. (2001) Aspartyl protease inhibitor pepstatin binds to the presenilins of Alzheimer's disease. Biochem. 40, 8359-836Greenough, M.A., Volitakis, I, Li, Q.-X., Laughton, K., Evin, G., Ho, M., Dalziel, A.H., Camakaris, J, Bush, A.I. (2011) Presenilins promote the cellular uptake of copper and zinc and maintain copper chaperone of SOD1-dependent copper/zinc superoxide dismutase activity. J. Biol Chem. 286, 9776-8

 1800 605-5127
1800 605-5127 +61 (0)8 8352 7711
+61 (0)8 8352 7711