Tyrosine Kinase Receptor B, phospho Ser478/479 (TrkB, pS478/479), Rabbit Polyclonal Antibody
- Product Name Tyrosine Kinase Receptor B, phospho Ser478/479 (TrkB, pS478/479), Rabbit Polyclonal Antibody
-
Product Description
Rabbit anti-Tyrosine Kinase Receptor B, phospho Ser478/479 (TrkB, pS478/479) Polyclonal Antibody (Unconjugated), suitable for WB, IHC-Frozen.
- Alternative Names GP145-TrkB; Neurotrophic tyrosine kinase receptor type 2; TrkB tyrosine kinase; Tropomyosin-related kinase B; pTrkB; phosphoTrkB; phospho-TrkB
- Application(s) IHC-Frozen, WB
- Antibody Host Rabbit
- Antibody Type Polyclonal
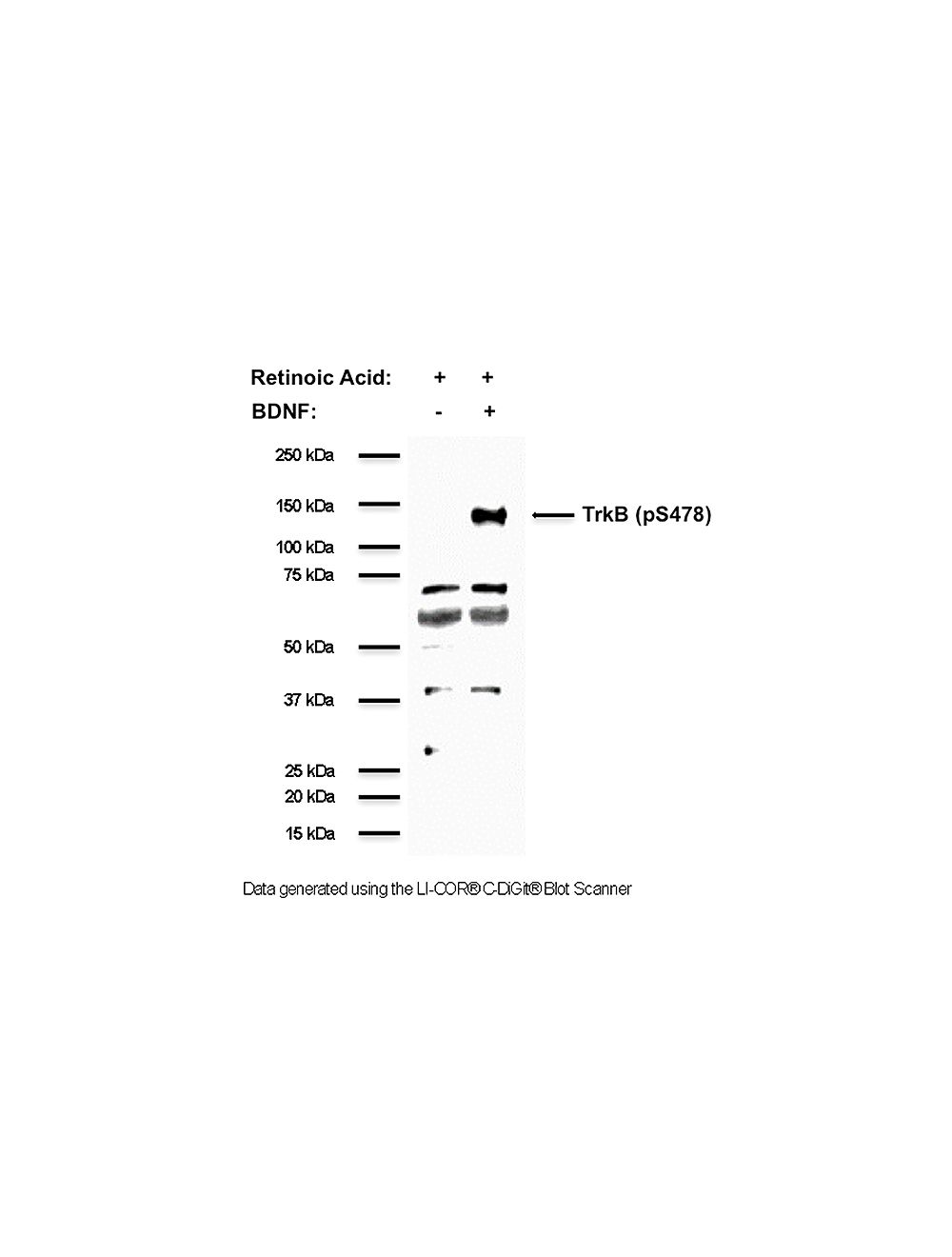
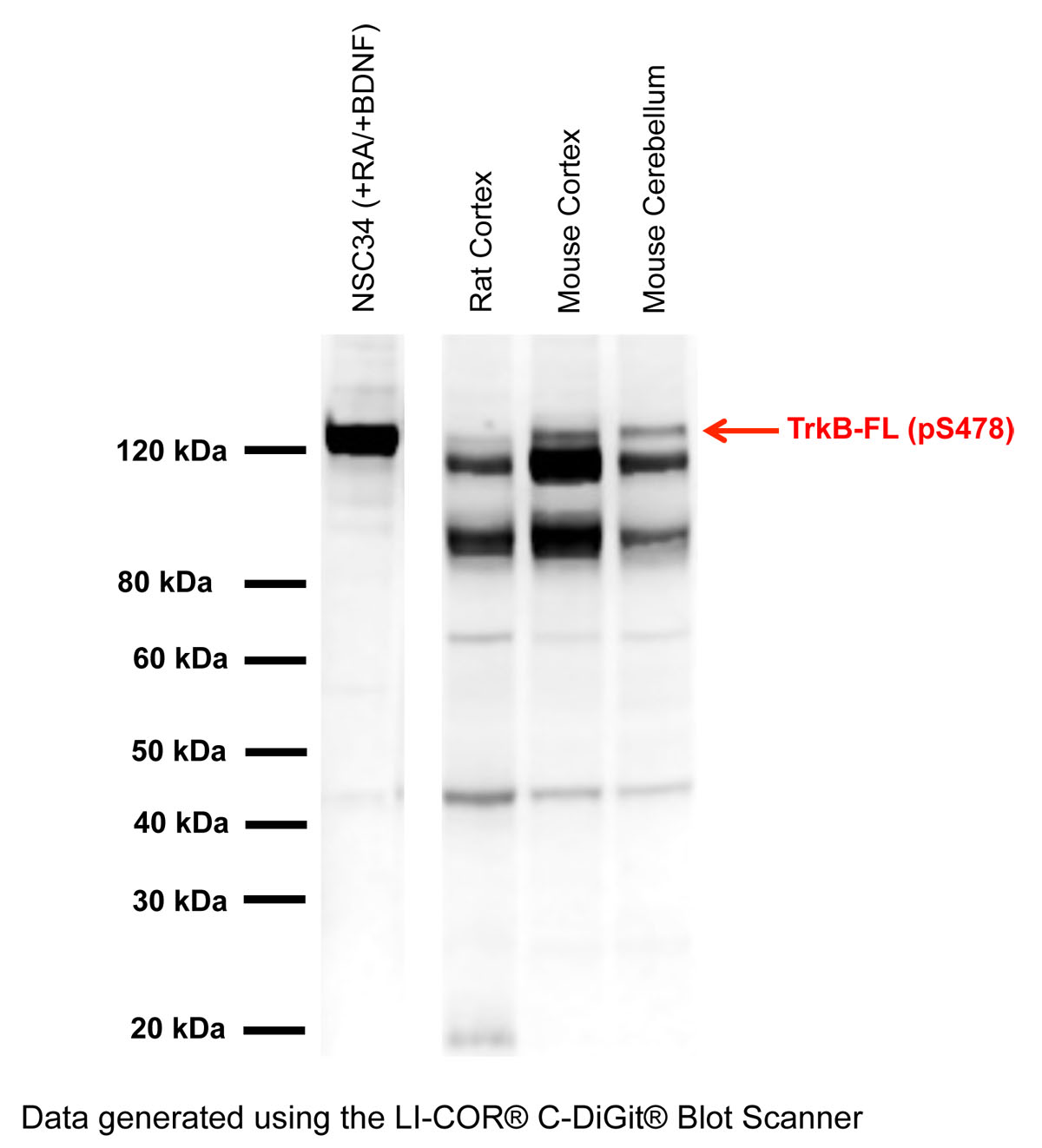
- Specificity Human TrkB (pS478). Antibody has been shown to be specific for TrkB phosphorylated on serine 478 by phospho-peptide absorption dot blots, and on cell lysates from cell lines induced with retinoic acid and BDNF. Antibody detects a clear band in retinoic acid (RA) and BDNF-treated NSC34 cell lysates at ~120 kDa only, demonstrating that the phosphorylated TrkB receptor is being detected. Additional non-specific bands at lower molecular weight may be observed in lysates and homogenates with the antibody and these bands have not been characterized.
- Species Reactivity Chicken (Predicted), Human, Mouse, Rat
- Immunogen Description Synthetic peptide immunogen, SNDDDSA[pS]PLHHIS
- Conjugate Unconjugated
- Purity Description Affinity purified, and absorbed.
- Regulatory Status For research use only.
Product Info
-
Product Description
Rabbit anti-Tyrosine Kinase Receptor B, phospho Ser478/479 (TrkB, pS478/479) Polyclonal Antibody (Unconjugated), suitable for WB, IHC-Frozen.
-
Related Products
TrkB (pS478) Positive Control Cell Lysate (Mouse NSC-34)
Tyrosine Kinase Receptor B (TrkB), Mouse Monoclonal Antibody
Tyrosine Kinase Receptor B, phospho Tyr816/Y817 (TrkB, pY816/817), Rabbit Polyclonal Antibody
Tyrosine Kinase Receptor B (TrkB), Rabbit Polyclonal Antibody
- Application(s) IHC-Frozen, WB
-
Application Details
Western Blotting (0.5 - 2 µg/mL). Cell lysates or membrane preparations prepared from isolated brain or spinal cord tissues are recommended. This antibody works on skim milk blocked membranes, however, best results are obtained with a equal mixture of 2.5% skim milk / 2.5% highly purified BSA as blocking reagent and antibody diluent.
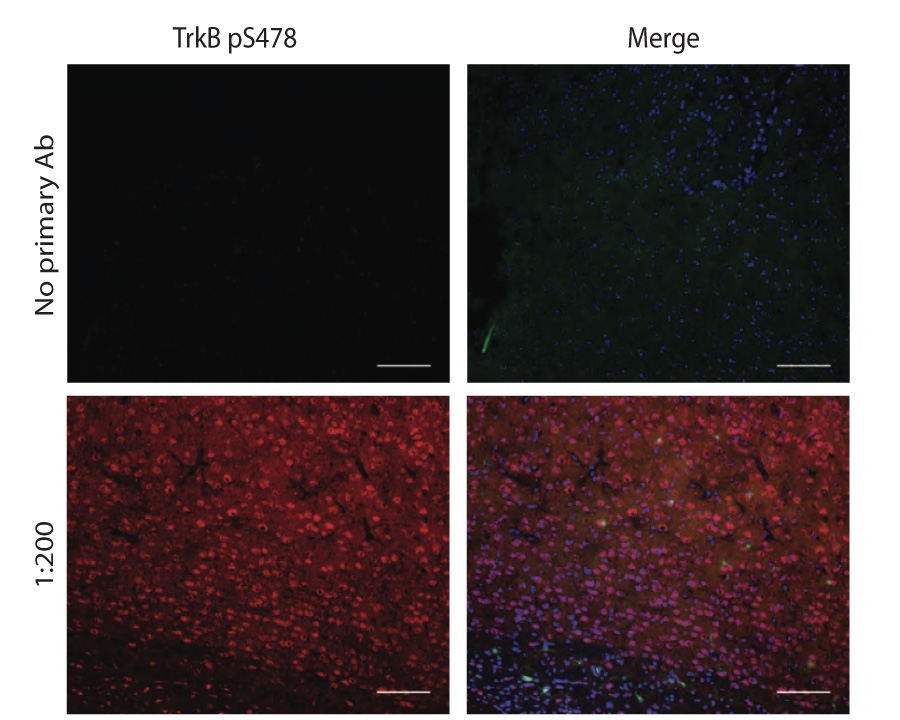
Immunohistochemistry (1 - 5 µg/mL). Antibody has been shown to work on PFA fixed, frozen sections. TBS is preferred for buffer preparation.
Other applications have not been tested. Biosensis recommends optimal dilutions/concentrations should be determined by the end user. - Product Validation Antibody is specific for TrkB serine phosphorylation at amino acid 478/479 (rodents/human). Lambda-phosphatase treatment obliterates positive staining.
- Target Tyrosine Kinase Receptor B, phospho Ser478/479 (TrkB, pS478/479)
- Specificity Human TrkB (pS478). Antibody has been shown to be specific for TrkB phosphorylated on serine 478 by phospho-peptide absorption dot blots, and on cell lysates from cell lines induced with retinoic acid and BDNF. Antibody detects a clear band in retinoic acid (RA) and BDNF-treated NSC34 cell lysates at ~120 kDa only, demonstrating that the phosphorylated TrkB receptor is being detected. Additional non-specific bands at lower molecular weight may be observed in lysates and homogenates with the antibody and these bands have not been characterized.
- Target Host Species Human
- Species Reactivity Chicken (Predicted), Human, Mouse, Rat
- Antibody Host Rabbit
- Antibody Type Polyclonal
- Antibody Isotype IgG
- Conjugate Unconjugated
- Immunogen Description Synthetic peptide immunogen, SNDDDSA[pS]PLHHIS
- Immunogen Length 14 amino acids
- Sequence SNDDDSA[pS]PLHHIS
- Epitope Intracellular (cytosolic)
- Positive Control Retinoic acid- and BDNF-treated NSC34 cells
- Isoform Information Detects full-length TrkB phosphorylated at amino acid S479 in humans (S478 in mouse and rat).
- Purity Description Affinity purified, and absorbed.
- Physical State Solid
- Format Lyophilized.
-
Formulation
Lyophilized from 1 x PBS, pH 7.2-7.6, with 0.1% trehalose
- Reconstitution Instructions Spin vial briefly before opening. Reconstitute in 50 µL sterile-filtered, ultrapure water. Centrifuge to remove any insoluble material. Final buffer contains no preservatives.
- Storage Instructions Store lyophilized antibody at 2-8°C. After reconstitution divide into aliquots and store at -20°C for long-term storage. Store at 2-8°C short-term (up to 4 weeks) with an appropriate antibacterial agent. Avoid repetitive freeze/thaw cycles.
- Batch Number Please see item label.
- Expiration Date 12 months after date of receipt (unopened vial).
- Alternative Names GP145-TrkB; Neurotrophic tyrosine kinase receptor type 2; TrkB tyrosine kinase; Tropomyosin-related kinase B; pTrkB; phosphoTrkB; phospho-TrkB
- Uniprot Number Q16620
- Uniprot Number/Name Q16620 (NTRK2_HUMAN)
- Cellular Localization Cell membrane
- Scientific Background The protein named TrkB (also named Neurotrophic tyrosine kinase receptor type 2 (NTRK2), GP145-TrkB or Tropomyosin-related kinase B is a receptor tyrosine kinase involved in the development and the maturation of the central and the peripheral nervous systems and is important in the regulation of neuron survival, proliferation, migration, differentiation, and synapse formation and plasticity. TrkB may also play a role in neutrophin-dependent calcium signaling in glial cells and mediate communication between neurons and glia. TrkB is the primary receptor for BDNF (brain-derived neurotrophic factor. TrkB also binds NT4 and NT3 but less efficiently. Upon ligand-binding, the receptor undergoes homodimerization, autophosphorylation and activation. TrkB activation recruits, phosphorylates and/or activates several downstream effectors including SHC1, FRS2, SH2B1, SH2B2 and PLCG1 that each regulate distinct overlapping signaling cascades within cells. Through SHC1, FRS2, SH2B1, SH2B2, these activate the GRB2-Ras-MAPK cascade that regulates, for instance, neuronal differentiation including neurite outgrowth. These same effectors also control the Ras-PI3 kinase-AKT1 signaling cascade that mainly regulates growth and survival. TrkB, via activation of PLCG1 and the downstream protein kinase C-regulated pathways, also controls synaptic plasticity, and thus plays a role in learning and memory by regulating both short term synaptic function and long-term potentiation. PLCG1 also leads to NF-Kappa-B activation and the transcription of genes involved in cell survival. One such consequence is that PLCG1 activation via TrkB is able to suppress anoikis, the apoptosis resulting from loss of cell-matrix interactions. (Reference: www.uniprot.org)
- Shipping Temperature 25°C (ambient)
- UNSPSC CODE 41116161
- Regulatory Status For research use only.
Specifications
-
Specific References
Turnbull MT et al. (2018), Acute Down-regulation of BDNF Signaling Does Not Replicate Exacerbated Amyloid-β Levels and Cognitive Impairment Induced by Cholinergic Basal Forebrain Lesion. Front Mol Neurosci. 2018 Feb 22;11:51. Species: Mouse; Application: WB, hippocampal lysates.
Matusica D et al. (2016), Inhibition of motor neuron death in vitro and in vivo by a p75 neurotrophin receptor intracellular domain fragment. J Cell Sci. 2016 Feb 1;129(3):517-30. doi: 10.1242/jcs.173864. Epub 2015 Oct 26. Species: Mouse; Application: WB, spinal cord lysates.
- General References Lai K-O. et al. (2012), Nature Neuroscience 15, 1506-1515, doi:10.1038/nn.3237.Zhao L. et al. (2009), Journal of Cell Science 122, 3123-3136, doi: 10.1242/jcs.047712.

 1800 605-5127
1800 605-5127 +61 (0)8 8352 7711
+61 (0)8 8352 7711